The 3rd Cell Therapy Analytical Development Summit Europe– What’s It About?
Stand at the forefront of analytical innovation at the 3rd Cell Therapy Analytical Development Summit Europe, the premier event for technical professionals - analytical development, quality control, CMC, Tech Ops, and more!
Learn how to stay ahead by integrating cutting-edge technologies such as ML anomaly detection and automation in assay execution. Navigating the analytical life cycle and surmounting regulatory hurdles are equally critical, to showcase the unique qualities of your ground-breaking product.
Don't miss out on this opportunity to hear from industry leaders, share your own insights and uncover ground breaking strategies to maximise product efficacy!
TAKE A LOOK AT THE FULL AGENDA
What Insights Could You Gain?
Highlight best practices for strategically placing control measures, emphasising continuous monitoring and adaptation to better address emerging risks with Tigen Pharma and Johnson & Johnson.
Ensure product safety and quality through machine learning for enhanced quality control and anomaly detection with GSK and real time monitoring with Avobis Bio for revolutionised quality control.
Design and craft cost effective assays for enhanced characterisation and streamlined regulatory compliance with Resolution Therapeutics.
Uncover regulatory insights for navigating successful IND/IMPD applications and advancing analytical methods in both clinical and commercial stages for accelerated development with regulatory experts.
Discover innovative approaches to classic methods including bypassing traditional methods for more accurate apoptosis assessments utilising real-time insights for point-of-care testing in cell therapy analysis and prioritising viability and functionality in storage methods with Tigen Pharma.
Who Will You Meet?
Connect with industry peers who share your passion for analytical development and are eager to explore the cutting-edge advancements. Get in touch here to discuss your networking opportunities further.
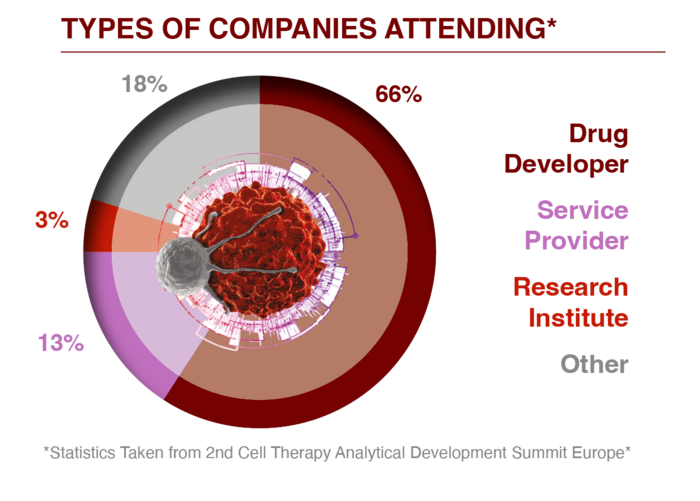
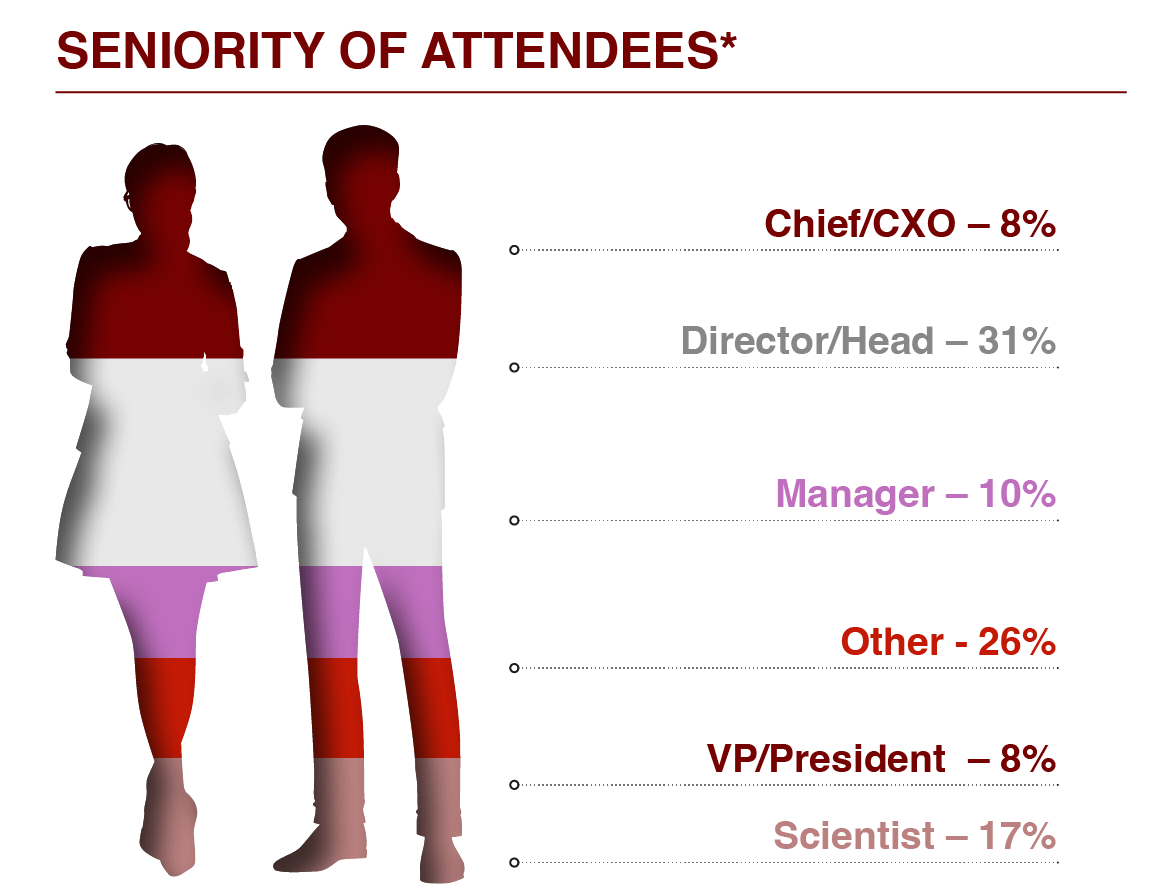
For further information or to access an attendee list please enquire here.
What Your Peers Have to Say:
"Cell therapy continues to change and expand the therapeutic field. To ensure the treatment is available, safe and potent, the analytical environment should coevolve."
– Amanda Versteilen, Associate Director, Quality Control Analytical, Bristol Myers Squibb (2024 Speaker)
“It is great to have a forum to discuss in depth, the technical and regulatory implications associated with analytical development and analytical change during development. I look forward to hearing about experiences as well as sharing my own. I learn something new every time I attend.”
– Sharon Lonhurst, Independent Expert (2024 Speaker)
